- In-Stock Tumor Cell Lines
- Human Orbital Fibroblasts
- Human Microglia
- Human Pulmonary Alveolar Epithelial Cells
- Human Colonic Fibroblasts
- Human Type II Alveolar Epithelial Cells
- Human Valvular Interstitial Cells
- Human Thyroid Epithelial Cells
- C57BL/6 Mouse Dermal Fibroblasts
- Human Alveolar Macrophages
- Human Dermal Fibroblasts, Adult
- Human Lung Fibroblasts, Adult
- Human Retinal Muller Cells
- Human Articular Chondrocytes
- Human Retinal Pigment Epithelial Cells
- Human Pancreatic Islets of Langerhans Cells
- Human Kidney Podocyte Cells
- Human Renal Proximal Tubule Cells
Background
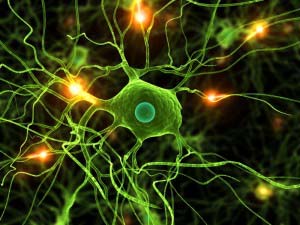 Microglia are a type of small macrophage-like glial cells in the central nervous system. Microglia can engage in phagocytosis and are involved in immune responses within and the development and remodeling of the nervous system. Their shape varies depending on the developmental and functional state, and anatomical location.
Microglia are a type of small macrophage-like glial cells in the central nervous system. Microglia can engage in phagocytosis and are involved in immune responses within and the development and remodeling of the nervous system. Their shape varies depending on the developmental and functional state, and anatomical location.
Microglia are the resident macrophages in the CNS and are ontogenically distinct from other phagocytic cells in the mononuclear phagocyte system such as dendritic cells, macrophages, and monocytes.
Microglia originate from primitive macrophages derived from erythro-myeloid precursors in the yolk sac. These cells colonize the brain during development and remain of embryonic origin in the healthy CNS.
In the steady-state condition, microglia can be identified using several common markers which they share with macrophages, such as CD11b, CD45, CD68, and CX3CR1. These cells also express specific markers such as P2RY12, which allow for their distinction from other cell types in the brain as well as peripheral immune cells.
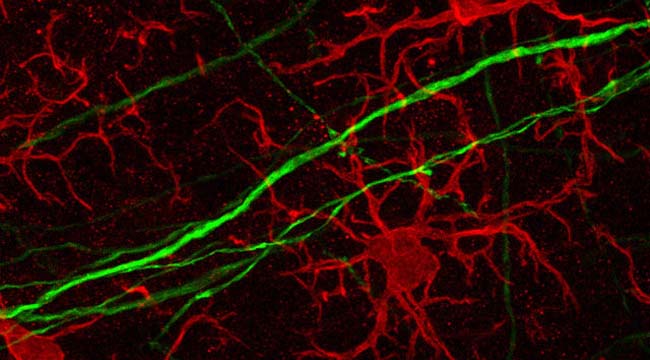
Fig1. Microglia Morphology
The Latest Research
-
1.Microglia and Alzheimer’s Disease
Many risk genes for the development of Alzheimer’s disease (AD) are exclusively or highly expressed in myeloid cells.
Microglia are dependent on colony-stimulating factor 1 receptor (CSF1R) signaling for their survival. The researchers designed and synthesized a highly selective brain penetrant CSF1R inhibitor (PLX5622), allowing extended and specific microglial elimination, both preceding and during pathology development.
The researchers found that in the 5xFAD mouse model of AD, plaques fail to form in the parenchymal space following microglial depletion, except in areas containing surviving microglia. Aβ deposits in cortical blood vessels reminiscent of cerebral amyloid angiopathy. Altered gene expression in the 5xFAD hippocampus is also reversed by the absence of microglia. Transcriptional analyses of the residual plaque-forming microglia show they exhibit a disease-associated microglia profile.
Researchers also described the structure, formulation, and efficacy of PLX5622, which allows for sustained microglial depletion and identifies the roles of neural cells in initiating plaque pathogenesis.
-
2.Microglia and Huntington’s Disease
Huntington’s Disease (HD) is a neurodegenerative disorder that manifests with movement dysfunction. The expression of mutant Huntingtin (mHTT) disrupts the functions of brain cells.
Galectin-3 (Gal3) is a lectin that has not been extensively explored in brain diseases. Herein, Siew.et al showed that the plasma Gal3 levels of HD patients and mice correlated with disease severity. Moreover, brain Gal3 levels were higher in patients and mice with HD than those in controls.
The up-regulation of Gal3 in HD mice occurred before motor impairment, and its level remained high in microglia throughout disease progression.
The cell-autonomous up-regulated Gal3 formed puncta in damaged lysosomes and contributed to inflammation through NFκB- and NLRP3 inflammasome-dependent pathways. Knockdown of Gal3 suppressed inflammation, reduced mHTT aggregation, restored neuronal DARPP32 levels, ameliorated motor dysfunction, and increased survival in HD mice.
Thus, suppression of Gal3 ameliorates microglia-mediated pathogenesis, which suggests that Gal3 is a novel druggable target for HD.
-
3.Microglia and Parkinson’s Disease
Cav1.2 channels are an L-type voltage-dependent Ca2+ channel, which is specifically blocked by calcium antagonists. Voltage-dependent Ca2+ channels are generally considered to be functional only in excitable cells like neural cells and muscle cells, but recently they have been reported to also be functional in non-excitable cells like microglia, which are principal players in the innate immune system and are involved in the pathophysiology of Parkinson’s disease.
Moreover, intensive degeneration of dopaminergic neural cells and accompanying behavioral deficits were observed in microglia-specific Cav1.2 knockdown mice intoxicated with MPTP, a neurotoxin that induces Parkinson’s disease-like symptoms, suggesting detrimental effects of microglial Cav1.2 blockade on Parkinson’s disease.
Therefore, the microglial Cav1.2 channel may have neuroprotective roles under physiological conditions and may also contribute to recovery from disease conditions.
Therefore, microglia are closely related to many major human diseases and it’s essential to study the function of microglia in the human body.
AcceGen provides high-quality human/animal microglia, including Human Microglia, Immortalized Human Microglia, CD-1 Mouse Microglia, CD57 Mouse Microglia, Immortalized Mouse Microglia, Rat Microglia and More. For more information please visit our website or contact inquiry@accegen.com.

Copyright - Unless otherwise stated all contents of this website are AcceGen™ All Rights Reserved – Full details of the use of materials on this site please refer to AcceGen Editorial Policy – Guest Posts are welcome, by submitting a guest post to AcceGen you are agree to the AcceGen Guest Post Agreement – Any concerns please contact marketing@accegen.com