Post Date:Mar-31-23
Improved CAR-T Cells May Eradicate Fatal Solid Tumors
 Author: AcceGen R&D Team
Author: AcceGen R&D TeamIntroduction
Chimeric Antigen Receptor (CAR)-T cells are modified T cells that carry single chain variable region (scFv) of an antibody coupled with T cell receptor (TCR) by gene transduction technology (Figure 1). The patient’s T cells were “reprogrammed” to generate a large number of tumor-specific CAR-T cells. Cancer immunotherapy by CAR-T cells has shown exhilarative clinical efficacy for hematological malignancies[1], however, challenges remain for use of CAR-T cell therapy to treat solid tumors, mainly for the limited tumor infiltration, host and tumor microenvironment, antigen escape, off-target effects and life-threatening toxicities.[2]
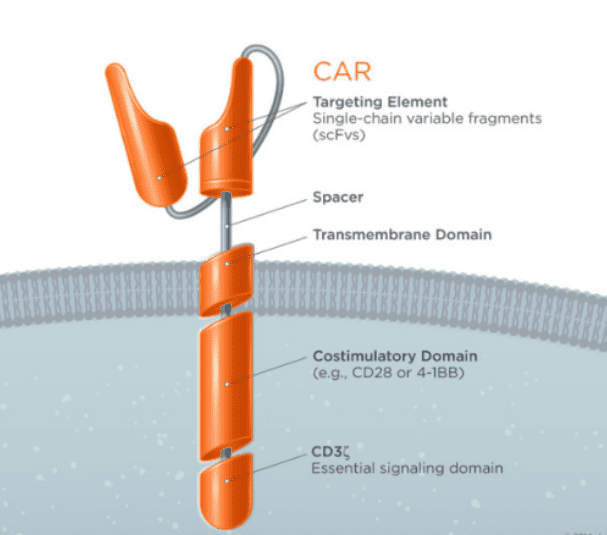
Figure 1. Basic structure of CAR-T[3]
Cell Culture
To prepare enough effective T cells for therapy, their rapid expansion is important. The commonly used T cell culture mediums in vitro include serum-containing media and serum-free media, namely RPMI 1640, IMDM, Gibco OpTmizer CTS T Cell Expansion SFM, Gibco AIM-V Medium CTS, LONZA X-VIVO 15, and StemSpan SFEM with or without Dynabeads Human T-Activator CD3/CD28. A study suggested that serum-free media provide a better environment for T cells to proliferate. Among the three serum-free media, scientists identify OpTmizer and AIM-V as better T cell culture environments than X-VIVO due to T cells being proved to have higher viability in the first two media. Besides, they found that in vitro human T cells keep a relatively resting status among non-CD3/CD28 groups, since they have weak proliferation and apoptosis abilities. [4]
The quality detection of CAR-T cells mainly includes safety, purity and potency detection. In addition to conventional endotoxin, mycoplasma, total anaerobe, yeast and mold, it is also necessary to ensure the high quality of the virus for the viral vector used for transduction. For the viral vector used for transduction, it is also necessary to ensure the high quality of the virus, in addition to conventional endotoxin, mycoplasma, total anaerobe, yeast and mold. Flow Cytometry is mainly used for the identification and purity detection of cell surface markers (CD3+, CD4+ or CD8+, CD62L+, CD45RA+) in CAR-T cells. ELISA detection kits are used to assess the potency of CAR-T cells.
The Latest Application of CAR–T Cells
Improving the invasion and infiltration capacity of CAR-T Cells, overcoming the tumor microenvironment, multiplexing CAR-T cells to target tumor profiles and reducing CAR-T cell toxicity are four effective strategies to overcome the problems mentioned above (Figure 2).
CAR-T cell trafficking requires the establishment of chemotactic migration between chemokines secreted by tumor cells and chemokine receptors on effector T cells. T cells manipulated with the chemokine receptor CXCR2, which binds to the ligand CXCL1 on tumor cells, have been demonstrated to effectively attack melanoma.[5] Anti-angiogenic therapy targeting VEGF or endothelin B receptor has been proved to normalize tumor vasculature and can be used in combination with targeted therapy similar to CAR-T cells to increase tumor infiltration [6,7].

Figure 2. Strategies to refine CAR-T cells for the treatment of solid tumors[1]
After the expression of CSF-1R in CAR-T cells, CAR-T cells showed a reaction to CSF-1 that enhanced proliferation signal transduction in CAR-T cells without affecting cytotoxicity[8]. Genetically engineered Gr-1 antibody exhausted myeloid-derived suppressor cells (MDSC) were used to improve the tumor-killing efficacy of CAR-T cells in mice[9]. Scientists broke down the physical barrier in solid tumors by constructing CAR-T cells that secreted ECM-degrading enzymes[10].
On December 16, 2022, in a study published in Science, scientists designed enhanced CAR T cells with a second receptor utilizing synthetic Notch receptors technologies. The second receptor recognized tumor antigens and subsequently caused a release of cytokine interleukin 2 (IL-2) from T cells. In the mouse models, these CAR T cells infiltrated solid pancreatic tumors and melanoma, resulting in substantial tumor eradication. A few solid tumors have no reaction to T-cell therapy for the immunosuppressive microenvironment preventing T-cell infiltration, activation, and proliferation. The major tumor inhibition mechanisms include inhibition of T cell receptor signaling and depletion of inflammatory cytokines. Local cell-based IL-2 circuits showed no toxicity due to the synNotch-IL-2 circuit targeting tumors independent of TCR/CAR activation. High levels of IL-2 can drive effective antitumor function of T cells with serious adverse effects, while autonomic cytokines are delivered directly to the tumor. The synNotch receptor drives the production of IL-2, and this tumor-targeting IL-2 offers a potential way to overcome local tumor inhibition and minimize the risk of the systemic use of IL-2. Immunoassay showed that CAR-T cells only expanded within the tumor, with increased activation markers and decreased depletion markers. The production of synthetic IL-2 allowed invasive T cells to survive and initiate continued CAR-mediated activation, amplification, and tumor killing. This synthetic cytokine delivery circuit could provide a powerful universal approach for remodeling and overcoming immunosuppressive solid tumors.[11]
In addition, CAR-T cells modified with specific T cell subsets may yield better results. CAR-T cells with high expression of CD26 secreted IL-17A, IFN-γ, IL-2, IL-22, and expressed CCR2 and CCR5 on the cell surface, showing the characteristics of memory stem cells. Universal CAR-T cell therapy is a prospective approach with a big challenge of GvHD. GvHD is associated with the HLA gene. Therefore scientists knocked down TCR-α or HLA using CRISPR-Cas9 technology[12], which meant CAR-T cells can be made from allogeneic cells. This could dramatically reduce the cost, time and resources required to generate CAR-T cells for every patient.
Recently Ahmad Khalil et al. developed a toolkit of 11 programmable synthetic transcription factors. These transcription factors can be activated by FDA-approved small molecule inducers to active cellular programs such as proliferative and antitumor activity.[13] Scientists tested the in vivo efficacy of Synziftr-regulated CAR T cells using a simple xenograft tumor model. The results show that the synZiFTR circuit can be used to program drug-dependent, post-delivery control of the antitumor activity of T cells in vivo. In addition to controlling CAR-mediated tumor targeting, synZiFTR was also suitable for controlling the expression of other proteins, such as IL-2 or Il-12, which can enhance the efficacy of immune cells by controlling these proteins to ensure safety.
Future Perspective
In solid tumors, getting CAR-T cells to attack and infiltrate the tumor is a challenge. This obstacle can be compounded by the immunosuppressive microenvironment of malignancies. Effective treatment also runs the risk of CAR-T cell-associated toxicities such as CRS and neurotoxicity. However, despite the challenges, new strategies and potential solutions continue to evolve and may provide a path forward for more effective and safer future therapies.
Where to Get CAR-T cell Custom Service for Your Research?
Immunotherapy like CAR-T has become a revolution in the area of cancer treatment. AcceGen is on target to be a reliable provider for immunotherapy, to relieve the burden of cancer treatment and to improve human health.
Currently, AcceGen offers the CAR-T cell custom service that targeting various biomarkers to fit your requirements. We continue to focus on the field of immunotherapy, especially CAR-T and to update our products.
It is our pleasure to help relative researches to move forward. All the products of AcceGen are strictly comply with international standards. For more detailed information, please visit our product portfolio or contact inquiry@accegen.com.
Reference
[1] Shuo Ma, Xinchun Li, Xinyue Wang, et al. Current Progress in CAR-T Cell Therapy for Solid
Tumors. International Journal of Biological Sciences. 2019; 15(12): 2548-2560.
[2] Robert C, Sterner and Rosalie M, Sterner. CAR-T cell therapy: current limitations and
potential strategies. Sterner and Sterner Blood Cancer Journal (2021) 11:69.
[3] https://www.junotherapeutics.com/our-science/car-technology/
[4] Hao Xu, Na Wang, Wenyu Cao, et al. Influence of various medium environments to in vitro human T cell culture. In Vitro Cellular & Developmental Biology-Animal. 54, 559–566 (2018).
[5] Kershaw MH, Wang G, Westwood JA, Pachynski RK, Tiffany HL, Marincola FM, et al. Redirecting migration of T cells to chemokine secreted from tumors by genetic modification with CXCR2. Hum Gene Ther. 2002; 13: 1971-80.
[6] Daenen LG, Shaked Y, Man S, Xu P, Voest E, Hoffffman R, et al. Low-dose metronomic cyclophosphamide combined with vascular disrupting therapy induces potent antitumor activity in preclinical human tumor xenograft models. Mol Cancer Ther. (2009) 8:2872–81.
[7] Yang J, Yan J, Liu B. Targeting VEGF/VEGFR to modulate antitumor immunity. Front Immunol. (2018) 9:978. doi: 10.3389/fifimmu.2018.00978
[11] Allen GM, Frankel NW, Reddy NR, et al. Synthetic cytokine circuits that drive T cells into immune-excluded tumors. Science. 2022 Dec16;378(6625):eaba1624.
[12] Ren J, Zhang X, Liu X, Fang C, Jiang S, June CH, et al. A versatile system for rapid multiplex genome-edited CAR T cell generation. Oncotarget (2017) 8:17002–11.
[13] Li HS, Israni DV, Gagnon KA, et al. Multidimensional control of therapeutic human cell function with synthetic gene circuits. Science. 2022 Dec 16;378(6625):1227-1234. doi:10.1126/science.
Copyright - Unless otherwise stated all contents of this website are AcceGen™ All Rights Reserved – Full details of the use of materials on this site please refer to AcceGen Editorial Policy – Guest Posts are welcome, by submitting a guest post to AcceGen you are agree to the AcceGen Guest Post Agreement – Any concerns please contact marketing@accegen.com